This website uses cookies so that we can provide you with the best user experience possible. Cookie information is stored in your browser and performs functions such as recognising you when you return to our website and helping our team to understand which sections of the website you find most interesting and useful.
Examples of Assays - Blood Assay Solutions
1.
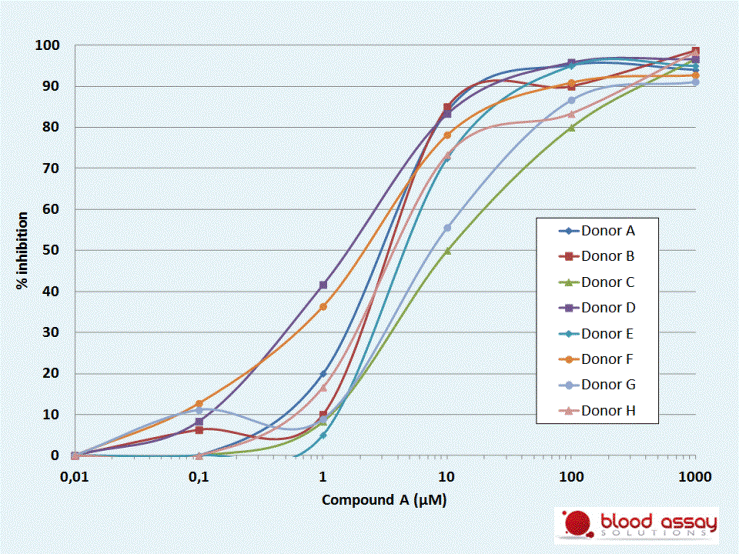
Compound A has been found to inhibit LPS-induced TNF release. IC50 will be measured in whole blood assays to assess donor to donor variation.
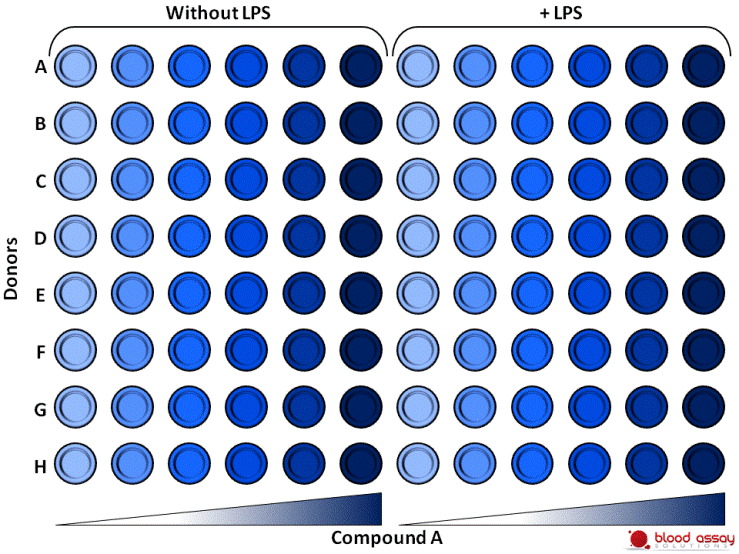
Assay design
Fresh whole blood from 8 different donors is preincubated with increasing concentrations of compound A and treated or not with LPS. After 4h incubation, TNF is measured in the supernatants.
Conclusion
Compound A inhibits LPS-induced TNF production in all the donors tested. IC50 ranges from 1.5 to 10µM.
2.
DETECTION OF
CONTAMINANTS
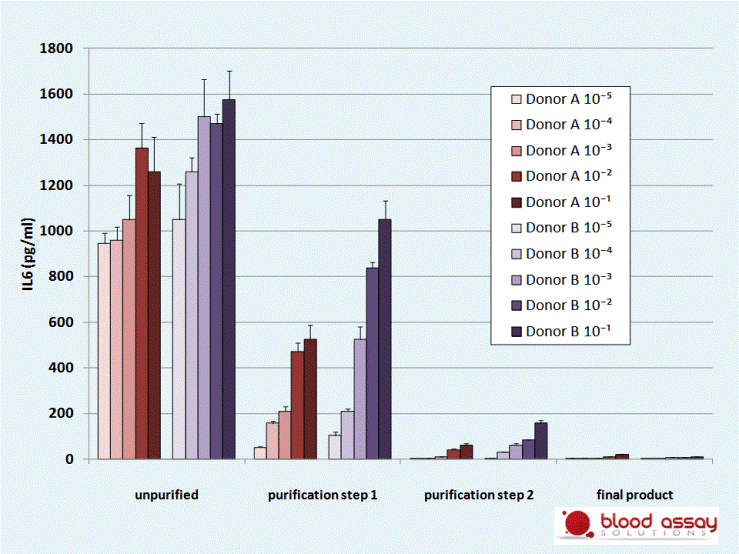
The purification of compound B comprises several decontamination steps to remove contaminants of bacterial origin. A whole blood assay is used to validate the removal of contaminants after each purification step.
Assay design
Fresh whole blood from 2 donors is incubated with serial dilutions of compound B sampled after each purification step. After 4h, IL6 is measured in the supernatants.
Conclusion
The final product is free of contaminants.
3.
COMPARISON OF DIFFERENT ADJUVANT FORMULATIONS
A vaccine company wishes to evaluate different formulations of an adjuvant.
Assay design
Fresh whole blood from 3 different donors is incubated 4-6h with serial dilutions of each formulation. The release of pro-inflammatory cytokine(s) (e.g. IL1, IL6, TNF) is then measured.
Conclusion
> Formulation #1 doesn’t induce a response in any of the donors.
> Formulation #3 is as efficient as the positive control.
> Formulation #4 is less efficient than formulation #3.
> Interestingly, formulation #2 works in donor A and C but not with donor B reflecting donor to donor variations specific to this formulation.